Associate Professor Sophie Payne, Head of Peripheral Interface Neuromodulation Research Program
Sophie is Head of the Peripheral Interface Neuromodulation Research Program at the Bionics Institute and is a Principal Research Fellow (honorary) at the University of Melbourne.
She completed her PhD in neuroscience at the University of Western Australia (2010-2013), then moved to the University of Melbourne (2013-2015) as a Research Fellow and later to the Bionics Institute (2015-present).
Using electricity to alter the activity of the vagus nerve has demonstrated success in the treatment of many human diseases.
Her research vision is to progress a pipeline of electric nerve technology towards the clinic to provide clinical relief in many inflammatory diseases. The vagus nerve stimulation technology is now in clinical trials for inflammatory bowel disease (ElectRx Study: NCT05469607) and rheumatoid arthritis (Reboot Study: NCT07017686).
More recently, Sophie has turned her attention to assess the efficacy of vagus nerve stimulation for the treatment of brain disorders such as epilepsy and Parkinson’s Disease.
Her Research Program has attracted $9.3M funding, and she has been recognised by the Australian Government and media as an emerging leader (Short-listed for the Prime Minister’s Science Award in 2023 & 2024; named as Australia’s ‘Top 25 female doctors and scientists in the Herald Sun & Daily Telegraph, 2023).
Sophie’s work in the field of ‘electric medicine’ has led to the publication of several world wide patents and over 30 papers.
Her work has also received media attention including featuring on Channel 7, 9, 10 tonight, ABC 24 evening news and Sunrise morning show, as well as several radio talks (ABC radio with Virginia Trioloi, 3AW, ABC Adelaide, Einstein-a-Go-Go radio) and Herald Sun articles (Brigid O’Connell).
Sophie has a presenter at the Museum’s Top Design Education Forum (2023) and an invited ‘exhibit scientist’ for the Melbourne Museum’s exhibition ‘A Gut Feeling’ during Science Week (2019).
Sophie is a current supervisor of 5 PhD students, has supervised >20 postgraduate students (Biomedical Research Victoria’s program), is a mentor for PhD and early career researchers (IMNIS) and ‘Mentoring the Next Generation of Women in STEMM’ program.
URL: https://www.bionicsinstitute.org/dr-sophie-payne
ORCID: 0000-0002-3428-2275
Google Scholar: Sophie Payne
Research projects
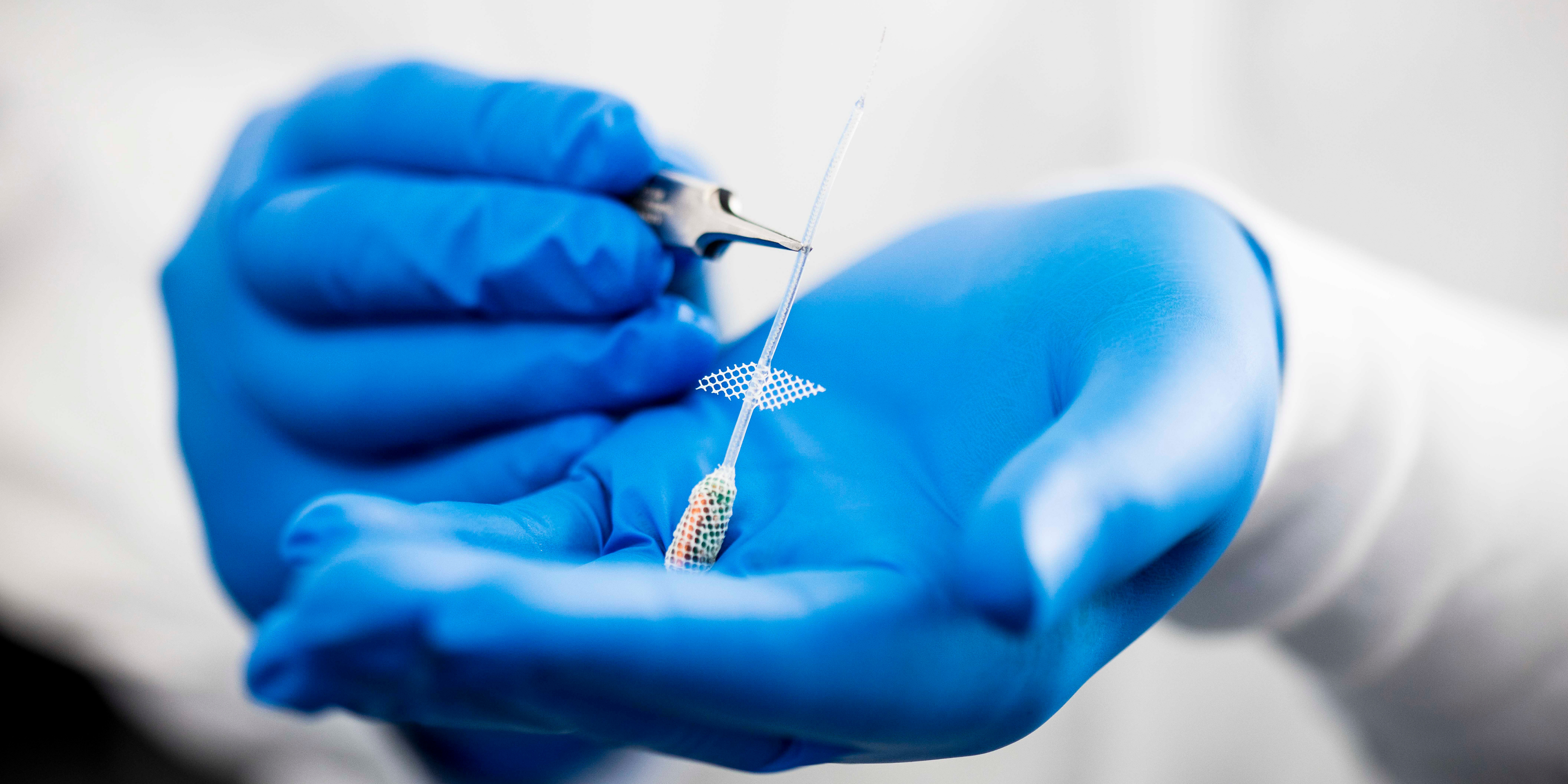
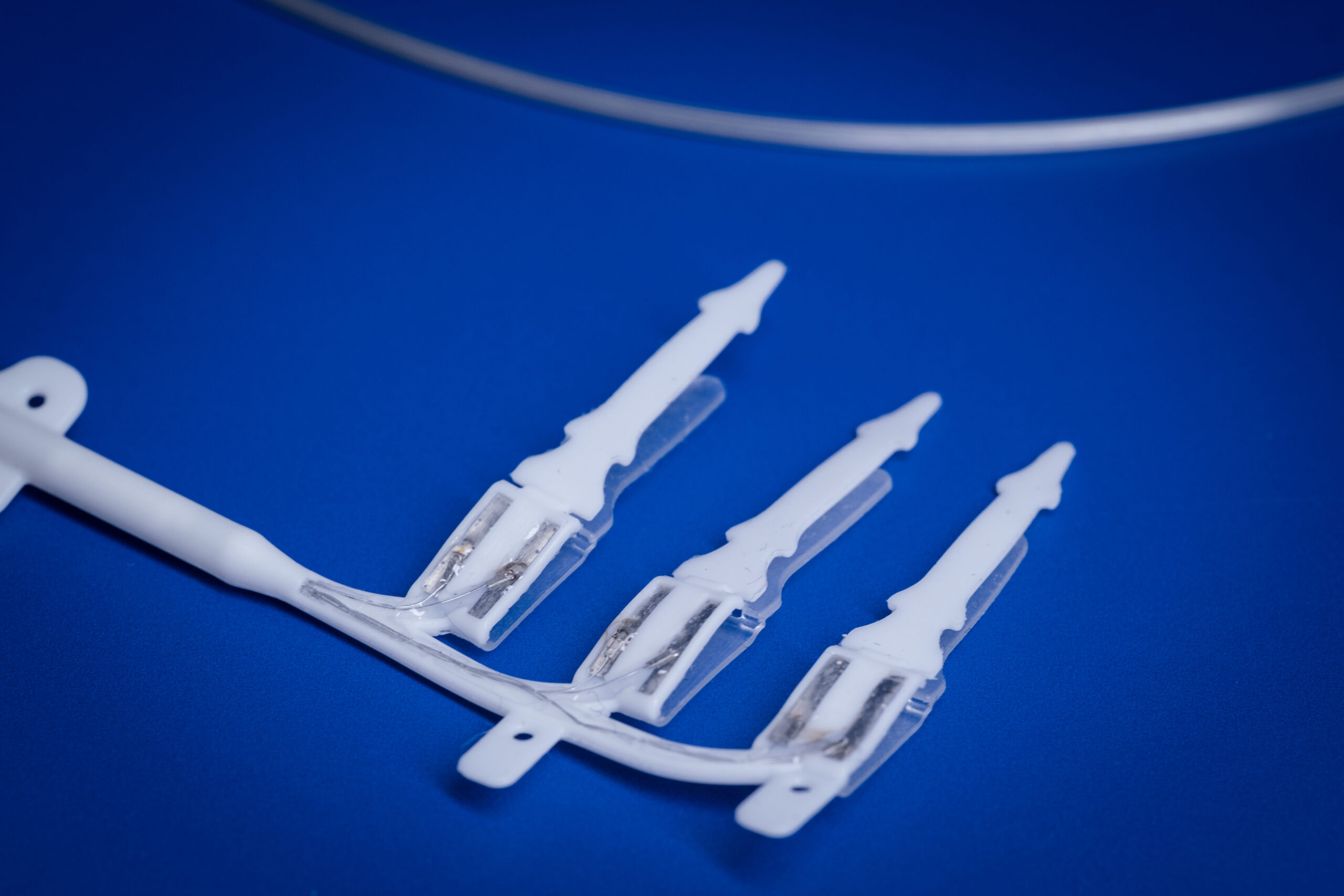
Miniaturised peripheral nerve device development
Closed-loop control of inflammatory bowel disease
Recent publications
1. Payne S.C, Wiedmann N.M, Eiber C.D, Wong A.W, Senn P, Osborne P.B, Keast J.R and Fallon J.B. 2020. Recording of electrically evoked neural activity and bladder pressure responses in awake rats chronically implanted with a pelvic nerve array. Frontiers in Neuroscience. 14:619275. doi.org/10.3389/fnins.2020.619275
2. Payne S.C, Ward G, MacIsaac R.J, Hyakumura T, Fallon J.B, Villalobos J. 2020. Differential effects of vagus nerve stimulation strategies on glycemia and pancreatic secretions. Physiological reports. 8(11): e14479. doi.org/0.14814/phy2.14479. Full Text
3. Payne, S. C., J. B. Furness, O. Burns, A. Sedo, T. Hyakumura, R. K. Shepherd, and J. B. Fallon. 2019. Anti-inflammatory Effects of Abdominal Vagus Nerve Stimulation on Experimental Intestinal Inflammation. Frontiers in Neuroscience. 13(418). doi.org/3389/fnins.2019.00418. Full Text
4. Payne, S. C., O. Burns, M. J. Stebbing, R. Thomas, A. C. de Silva, A. Sedo, F. Wiessenborn, T. Hyakumura, M. Huynh, C. N. May, R. A. Williams, J. Furness, J. Fallon, and R. Shepherd. 2019. Vagus nerve stimulation to treat inflammatory bowel disease: a chronic, preclinical safety study in sheep. Bioelectronics in Medicine. 1(4): 235-250. doi.org/10.2217/bem-2018-0011.
5. Payne, S. C., J. B. Furness, and M. J. Stebbing. 2018. Bioelectric neuromodulation for gastrointestinal disorders: effectiveness and mechanisms. Nature reviews. Gastroenterology & hepatology. 16(2): 89-105. doi.org/10.1038/s41575-018-0078-6 . Full Text